Novel microglial calcium signal is a rapid sensor of neuronal damage in vivo
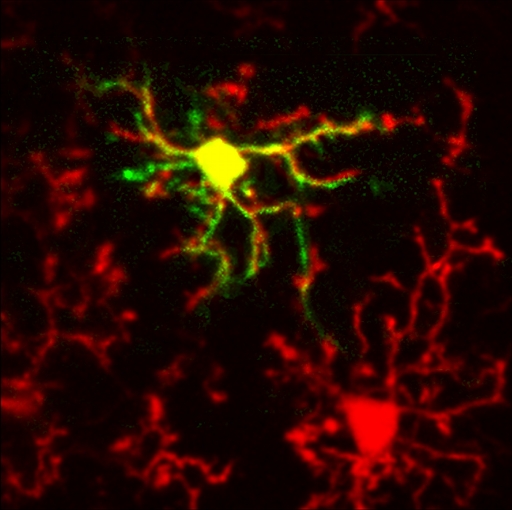 In the healthy adult brain microglia, the main immune-competent cells of the CNS, have a distinct (so-called “resting”, see image) phenotype. Resting microglia can only be studied in vivo since any isolation of brain tissue inevitably triggers microglial activation. Using in vivo two-photon imaging we obtained a first direct insight into Ca2+ signaling in resting cortical microglia.
In the healthy adult brain microglia, the main immune-competent cells of the CNS, have a distinct (so-called “resting”, see image) phenotype. Resting microglia can only be studied in vivo since any isolation of brain tissue inevitably triggers microglial activation. Using in vivo two-photon imaging we obtained a first direct insight into Ca2+ signaling in resting cortical microglia.
We have found that the majority of microglial cells show no spontaneous Ca2+ transients at rest and in conditions of strong neuronal activity. However, they reliably respond with large, generalized Ca2+ transients to damage of an individual neuron. These damage-induced Ca2+ transients (DICTs) have a short latency (0.4-4 s) and occur only in microglial cells localized to the immediate vicinity of the damaged neuron (< 50 µm cell body-to-cell body distance). DICTs are occluded by the agonists of metabotropic P2Y receptors and require Ca2+ release from the intracellular Ca2+ stores.
 Microglial calcium signal acts as a rapid sensor of single neuron damage in vivo
Microglial calcium signal acts as a rapid sensor of single neuron damage in vivoContact us for reprint.
The functional role of these long-lasting, generalized microglial Ca2+ signals remains to be explored. DICTs occur immediately after neuronal damage (on the millisecond-to-second scale) and thus long before the damage-induced changes in microglial morphology (initiated within the first few minutes) take place. Therefore DICTs most likely operate as a switch, triggering processes relevant to microglial activation. Such processes may include (i) reorganization of actin cytoskeleton, (ii) CREB phosphorylation, (iii) gene expression and de novo protein synthesis (e.g. production of cytokines and chemokines, cyclooxygenase-2 and BDNF), (iv) release of pro- and/or anti-inflammatory mediators and (v) microglial phagocytosis. The fact that DICTs remain restricted in space suggests that single-cell damage is treated as a local event, involving only immediately adjacent microglia.
Taken together our data suggest that in vivo somatic Ca2+ signaling in microglia is not involved in the surveillance of the extracellular milieu or in the detection of physiological levels of neuronal/astroglial activity. Rather, it functions as highly sensitive and specific signal for recognition of the damage in the microglial microenvironment.
- 78 views