Sleep spindles promote hippocampal network downregulation during sleep
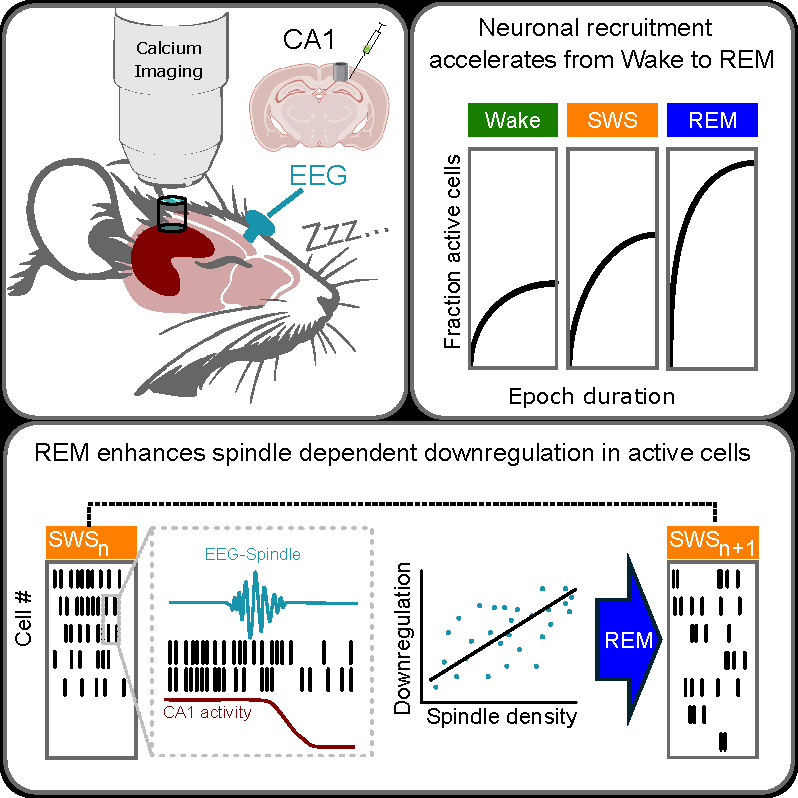 Sleep is thought to globally downregulate neuronal network activity and synaptic connections enhanced during prior wakefulness, and in parallel, to upregulate activity in networks mediating consolidation of hippocampus-dependent episodic memory. To assess these processes in hippocampal networks during natural sleep, we combined two-photon Ca2+ imaging of CA1 neuronal activity in mice with EEG recordings of sleep slow oscillations (SO) and spindles as markers of ongoing memory processing during slow wave sleep (SWS). We found that the number of active hippocampal neurons increased from wakefulness into sleep, peaking during rapid eye movement (REM) sleep. At the population level, Ca2+signaling did not decrease during individual SWS or REM epochs. Instead, we observed a gradual, persistent downregulation of Ca2+signaling across consecutive SWS epochs, specifically in neurons active during the preceding SWS epoch. In cells active during SOs and spindles, this downregulation was particularly pronounced when the SWS epochs were separated by a REM sleep epoch. Sleep spindle density during the preceding SWS epoch was the strongest predictor of this across-epochs downregulation. Moreover, spindle onsets were followed (within 1 s) by an immediate reduction in both the fraction of active cells and their mean Ca2+ transient amplitude. Our findings point to a spindle-associated mechanism that, in cooperation with intermittent REM sleep, drives the progressive downregulation of hippocampal network activity across sleep, potentially supporting systems memory consolidation.
Sleep is thought to globally downregulate neuronal network activity and synaptic connections enhanced during prior wakefulness, and in parallel, to upregulate activity in networks mediating consolidation of hippocampus-dependent episodic memory. To assess these processes in hippocampal networks during natural sleep, we combined two-photon Ca2+ imaging of CA1 neuronal activity in mice with EEG recordings of sleep slow oscillations (SO) and spindles as markers of ongoing memory processing during slow wave sleep (SWS). We found that the number of active hippocampal neurons increased from wakefulness into sleep, peaking during rapid eye movement (REM) sleep. At the population level, Ca2+signaling did not decrease during individual SWS or REM epochs. Instead, we observed a gradual, persistent downregulation of Ca2+signaling across consecutive SWS epochs, specifically in neurons active during the preceding SWS epoch. In cells active during SOs and spindles, this downregulation was particularly pronounced when the SWS epochs were separated by a REM sleep epoch. Sleep spindle density during the preceding SWS epoch was the strongest predictor of this across-epochs downregulation. Moreover, spindle onsets were followed (within 1 s) by an immediate reduction in both the fraction of active cells and their mean Ca2+ transient amplitude. Our findings point to a spindle-associated mechanism that, in cooperation with intermittent REM sleep, drives the progressive downregulation of hippocampal network activity across sleep, potentially supporting systems memory consolidation.
The article is in press at Curr. Biol.